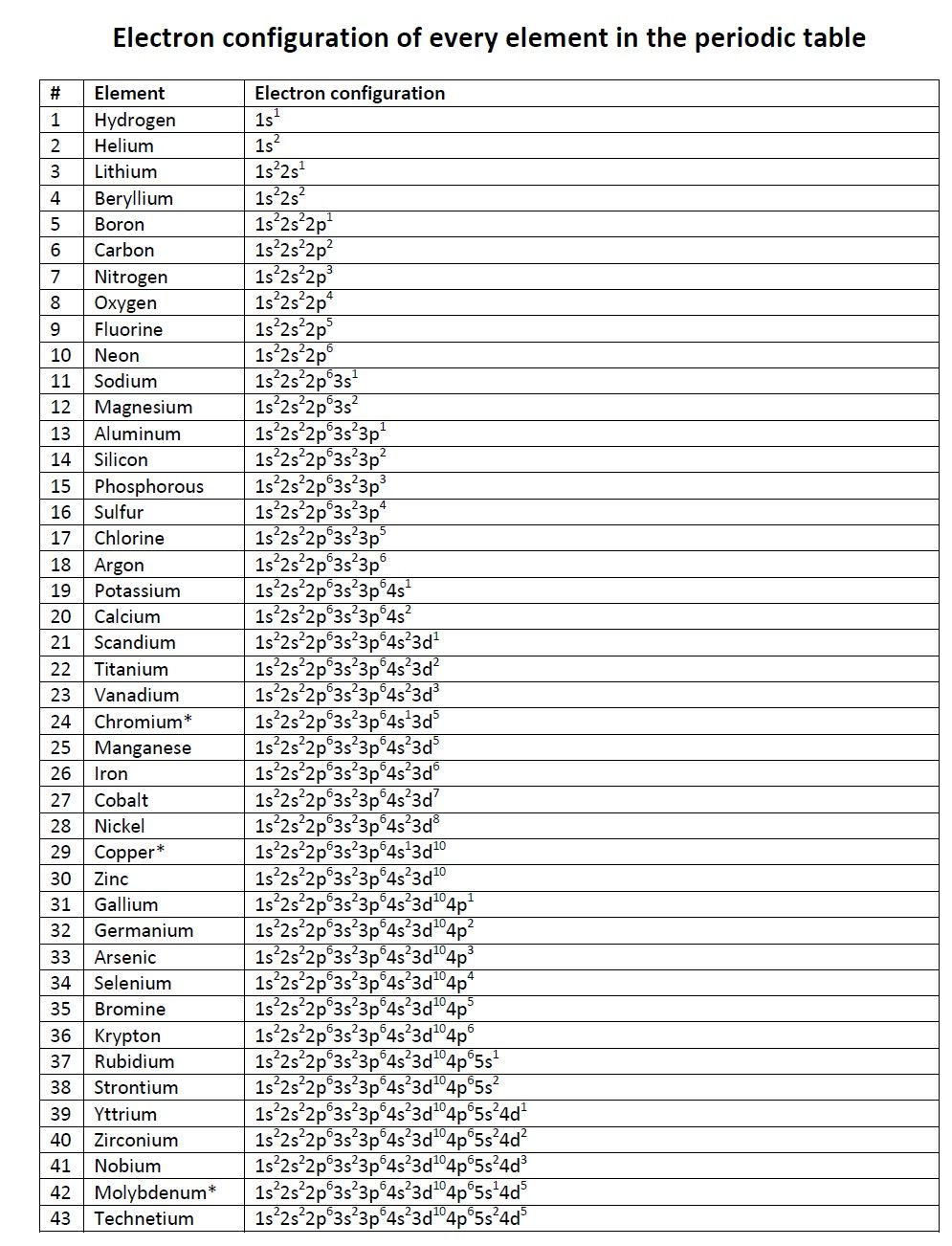
The general electronic configuration of valence electron = ns 1, where n = 1 to 7. Group-1 or IA in the periodic table contains seven elements like hydrogen, lithium, sodium, potassium, rubidium, cesium, and francium. Electronic configuration of group-1 elements Group-1 and 2 belong to s-block elements in the periodic table with general electron configuration ns 1→2, where n = number of electronic shells or the number of periods in which the element is present. Electronic configuration of s block elementsįor s-block elements, the electron enters the ns-orbitals and is progressively filled with atomic number. According to electronic configuration, the periodic table elements are classified into four blocks s, p, d, and f-block elements. The electron configuration formula of elements must be connected with the periodic table. Modern periodic tables are classified on the basis of chemical behavior and the electronic configuration of elements. The electronic configuration formula uses to derive some basic properties like the electromagnetic spectrum, chemical bonding, electric polarization, dipole moment, hydrogen bonding, etc. Electronic configuration and periodic table Spin pairing occurs only when vacant orbitals of similar energy are not available for occupation. What is Hund’s rule?Īccording to Hund’s rule, electrons are filling in the orbital with maximum spin multiplicity. Electrons with similar spin are configured first. The electron will tend to form maximum spin. 
The orbitals with the lowest energy filled up first while the highest energy orbital filled up in the end. According to this principle, the electrons are filled up in order of energy. 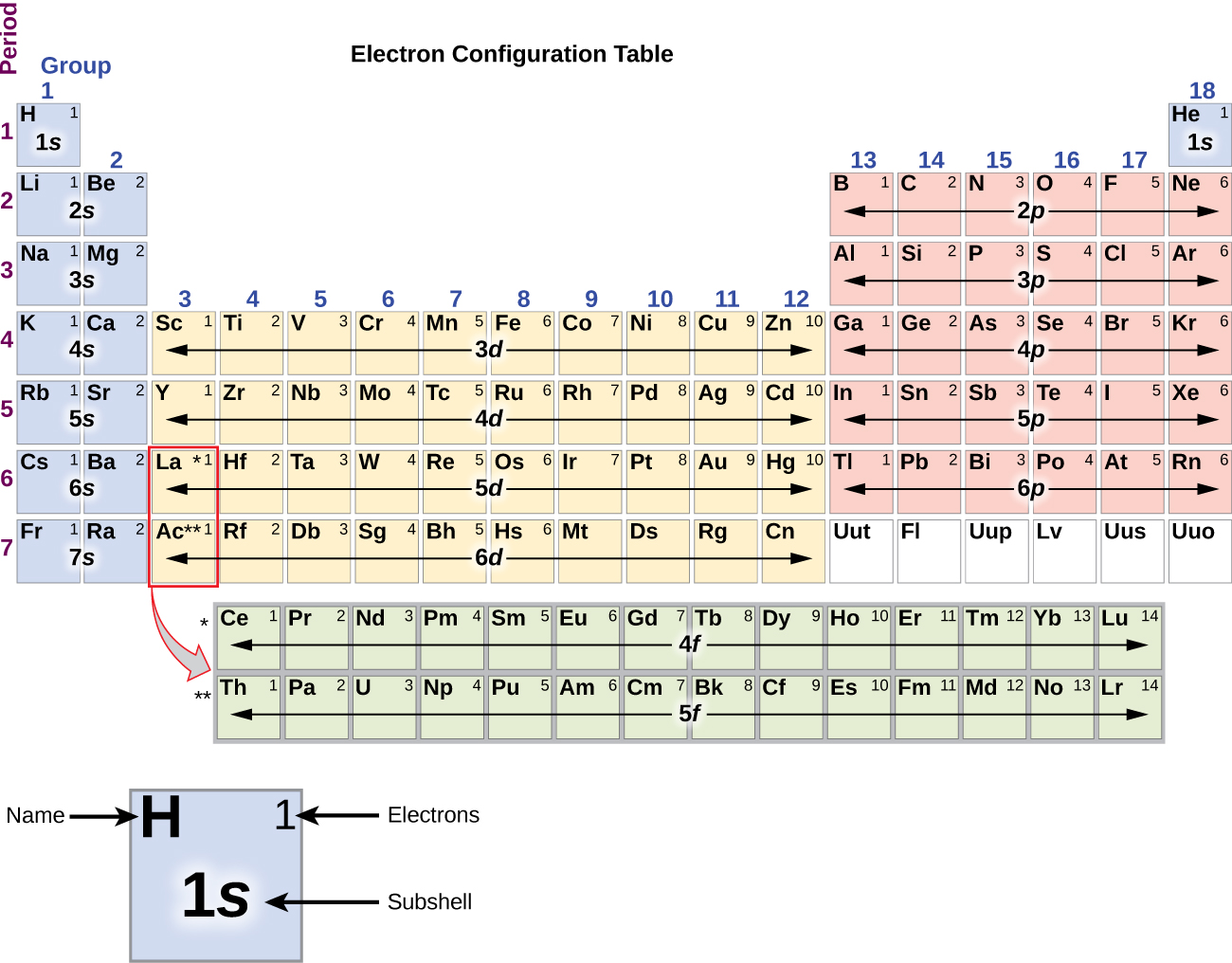
German scientist Aufbau expresses the building up principle for the electron configuration process in different electronic orbitals of atoms. Therefore, s, p, d, f energy levels have a maximum of 2, 6, 10, 14 electrons respectively.Īufbau principle and electron configuration Where l = 0, 1, 2, 3 for s, p, d, f orbitals. The maximum number of the electron in sub-shell like s, p, d, and f orbitals = 2(2l + 1). The maximum number of electrons in the main energy levels = 2n 2, where n = principal quantum number. The filling up electronic orbitals with electrons around the nucleus of atoms takes place according to certain rules. According to above diagram structure, configuration energy levels with electron constructed the following ordering, 1s < 2s < 2p < 3s < 3p < 4s < 3d < 4p < 5s < 4d < 5p < 6s < 4f < 5d < 6p < 7s < 5f…
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
